Reaction Dynamics
When a reaction begins, the reactants are consumed and the products are created. Thus, the concentration of the reaction decreases as well as the forward rate of reaction also decreases.
If the products are not allowed to escape (release from gas formation, etc.) then the products can react to re-form some of the reactants. When the reactant concentration increases, the reverse rate of reaction decreases.
This occurs when reactions are reversible, and they move in the forward and reverse directions.
Equilibrium relates to the reactions being able to move in the forward and backwards direction, and when equilibrium is reached the rate of products turning into reactants is the same rate of reactants turning into products.
WARNING: EQUILIBRIUM DOES NOT MEAN THE CONCENTRATIONS OR THAT THERE IS AN EQUAL AMOUNT OF PRODUCTS VERSES REACTANTS!
These reactions can have most of the reactants consumed when they are at equilibrium, and is defined by the using the arrow configuration below with a longer arrow pointing towards the products, and a smaller arrow pointing towards the reactants.
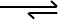
A reaction could also favour the reactants more than the products. This is seen in organic chemistry reactions, and methods such as Grignard Reagents are used to combat this issue, but that’s a future topic so stay tuned! Staying on topic, Only a small percentage of the reactant molecules are consumed at equilibrium, and is defined with a shorter arrow pointing towards the products, and the longer arrow pointing towards the reactants.
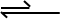
Dynamic Equilibrium
Dynamic Equilibrium: is the condition wherein the rates of the forward and reverse reactions are equal.
When the forward reaction, where the reactants turn into products, slows; the reverse reaction, where the products turn into products, accelerates.
Once the product reaches equilibrium, the concentrations of all the chemicals remain constant. This occurs because the chemicals are being consumed and made at the same rate of reaction.
Equilibrium Constant
There is a relationship between reactants and products at equilibrium, and this relationship is the called the Law of Mass Action.
A general equation for the Law of Mass Action
aA(aq) +bB(aq); ⇌ cC(aq) +dD(aq).
- The lowercase letters are representative of the coefficients of the balanced chemical equation, seen in the how to balance chemical coefficients post.
K is the letter used to represent the equilibrium constant, a unit-less dimension.
To calculate K the products are multiplied to the power of the coefficients divided by the reactants which are also raised to their corresponding coefficients.
K= [C]c x [D]d /[A]a x [B]b
An example of this will is 2 N2O5 (g) ⇌ 4 NO2 (g) + O2 (g) The equation to calculate the equilibrium constant of this reaction is
K= K= PNO24 x PO21 / P N2O52
Equilibrium Constant Expression
K=Kc is the Equilibrium constant for reactions only with aqueous components (aq), and is typically expressed with units of mol/L
K=KP is the Equilibrium constant for reactions only with gaseous components (g), and is typically expressed with units of bar
When a reaction contains both aqueous and gaseous components K is neither Kc nor KP
Essentially the Keq can determine the direction of the rate of the reaction.
When Keq is >> 1 the products are favoured, and when Keq is << 1 the reactants are favoured.
When the reaction is written backwards, for example when aA(g) + bB(g) ⇌ cC (g) + dD (g) becomes in the backward direction as follows cC (g) + dD (g) ⇌ aA(g) + bB(g) then Kforward= [C]c x [D]d /[A]a x [B]b becomes inverted as a results the reactants go to the top, and the products go to the bottom of the equation Kbackward=[A]a x [B]b/ [C]c x [D]d
When the coefficients of an equation are multiplied by a factor, the equilibrium constant is raised by that factor.
Heterogenous Equilibrium
Pure solids and pure liquids are materials whose concentrations doesn’t change (very much) during the course.
That means when constructing an ICE table, and that the mass or amount can change of the substance, however, the concentration does not.
Since this is the case, and their concentrations do not change the equation for the equilibrium constant does not include any pure solids or liquids.
aA(s) + bB(aq) ⇌ cC(l) + dD(aq) has the equilibrium constant expression Kc=[D]d/[B]b
No Kp expression if one of the reactants or products is aqueous
Calculating Equilibrium Constants from Measured Equilibrium Concentrations
The most direct way of finding the equilibrium constant is to measure the amounts of the reactants and products in a mixture at equilibrium.
The equilibrium value will always have the same constant, regardless of the differing amounts of products and reactants in the equilibrium mixture. This remains true as long as the temperature is kept constant, and the value of the equilibrium constant is independent of the initial amounts of reactants and products.
The K constant can be calculated using an ICE table.
Initial – gives the initial concentrations of the reactants and the products
Change – gives the change in value of the equilibrium
Equilibrium – The concentration of the reactants and the products once equilibrium has been reached, this is what will derive the equilibrium constant.
Stoichiometry can be used to determine the equilibrium concentrations of all reactants and products if you know initial concentrations and one equilibrium concentration. By using this change in concentration, the concentrations of the chemicals at equilibrium can be determined.
The Reaction Quotient
When the reaction mixture is not at equilibrium, we can determine the way the reaction will proceed by determining the Reaction Quotient. The equation for the reaction quotient is the determined the same way as the equilibrium constant. However, instead of using K, we use Q which means the reaction quotient. Q= [C]c x [D]d /[A]a x [B]b
Q is then compared with the equilibrium constant (Keq). When compared, if Q is greater than (>) Keq then the reaction will proceed towards the reactants (increases) rather than the products (decreases) (i.e. the reverse direction), if Q is less than (<) Keq then the reaction will proceed towards the products (increases) rather than the reactions (decreases) (i.e. the forward direction). Finally, if Q is approximately equal to Keq than the reaction.
If there are only reactants in a reaction mixture then Q=0, and then the reaction goes in the forwards direction. If there are only products then Q=∞, and then the reaction goes in the reverse direction
Keq is fluctuant, meaning that the equilibrium can be obtained with a greater amount of product or a greater amount of reactant.
Remember: Equilibrium does not mean equal amounts of products and reactants, but the an equal rate of reaction.
Directions: Finding the Equilibrium Concentrations
When you are given the equilibrium constant, and the initial concentrations, its is possible to find the equilibrium concentrations using basic algebra.
The first step is to decide which way the reaction is proceeding. This is done by comparing the Q and Keq values.
The second step is to define the changes of all the materials in terms of x. We do this by creating an ICE table.
- Use the coefficient from the chemical equation as the coefficient of x
- The x change is + for materials on the side of the reaction it is proceeding towards
- The x change is – for materials on the side of the reaction it is proceeding away from
The third and final step is to solve for x
- for 2nd order equations, take square roots of both sides or use the quadratic formula.
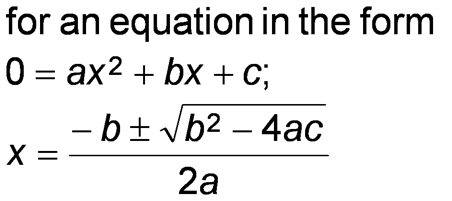
- It may be able to simplify and approximate the answer, rather than solving a complex quadratic equation. This is done for very large or small equilibrium constants.
How To: Simplify and Approximate
When the equilibrium constant is very small, the position of the equilibrium favours the reactants.
When the initial concentrations of the reactants are fairly large, the reactant will not change significantly when equilibrium is reached.
[X]equilibrium = ([X]initial – ax) ≈ [X]initial
With this equation the equilibrium concentration is approximated so that it is the same as the initial concentration. We are also assuming that the reaction is proceeding in the forward direction.
We can check our approximation by comparing the approximate value of Δx to concentrations which we chose to remain unchanged.That means we take divide Δx by the initial concentrations and then multiply by 100%. If the value of Δx is less than 5% the x value is valid, invalid if not.
A small K value is thought to be below 10-4 and a large K value is larger than 104 If K is in between 10-4 and 104 then x must be derived from the equilibrium problem. If K has a large value, then you must turn the products to zero by making x the size of the limiting reagent so that the reaction will become in the forward direction.
How to Disturb and Restore Equilibrium
Products and Reactant concentrations remain at equilibrium, however, this could be done by changing conditions. After changes are made to the condition the concentrations of all the chemicals will change until equilibrium is again restored.
Unless the temperature is changed, the new concentrations will be different, but the equilibrium constant will remains the same.
Le Châtlier’s Princple
Le Châtlier’s Principle says that if a system at equilibrium is disturbed, the position of equilibrium will shift to minimize the disturbance.
It guides us in predicting the effect of various changes in conditions, and the impacts they will have on the position of equilibrium.
The Effect of Catalysts
Catalysts provide an alternative more efficient mechanism.
Catalysts work for both forward and reverse reactions
Catalysts affect the rate of the forward and reverse reactions by the same factor. Thus, catalysts do not affect the position of equilibrium.
Leave a comment