Arrow Conventions
There are four different arrows commonly used in chemical equations.
1) The Single Arrow
The single arrow, you guessed it, is only one arrow. This arrow is pointed in one direction pointing from the reactants (the materials that are used) to the products (the product which is formed). This arrow indicates that pretty much all the reactants have been converted into products.
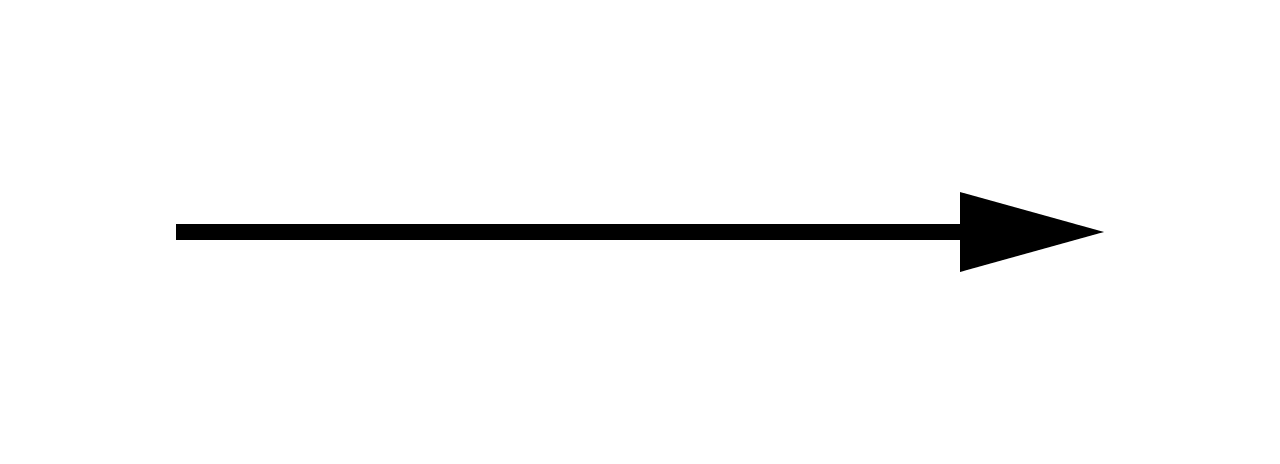
2) The Double Arrow
The double arrow, wow I think you might be a genius because you are right again, has two arrows. But it’s a little more complicated than you may think. There are a few variations of this arrow, and they all mean different things. Essentially what they mean is that a reaction becomes at equilibrium. Equilibrium will be discussed more further down, however, equilibrium does not mean that there are equal amounts of products or reactants, but there are
Top Arrow Longer Than Bottom Arrow
When the top arrow is longer than the bottom arrow, it means the the products are mostly favoured than the reactants. Which means that the reaction is going to drive more towards producing the products rather than producing the reactants. To remember this when you see the arrow with more, that means there is more of what the arrow is pointing at.

Bottom Arrow Longer Than Top Arrow
When the bottom arrow is longer than the top arrow, it means the the reactants are mostly favoured than the products. Which means that the reaction is going to drive more towards producing the reactants rather than producing the products.
To remember this when you see the arrow with more, that means there is more of what the arrow is pointing at.
This doesn’t mean that there is greater amounts of reactants than products that are being formed, but that there is an higher rate of reaction for the reactants than the products.
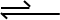
When the bottom arrow is equal than the top arrow, it means the the reactants are equally as favoured as the products. Which means that the reaction is going to drive towards equally producing reactants as producing the products.
To remember this when you see the arrow with the equal arrow length, that means there is equal conversion rate of both.
This doesn’t mean that equal amounts of products and reactants are being formed, but that there is an equal rate of reaction for both the products and reactants.
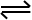
Leave a comment